Enabling NGS-Based Product Characterization and Biosafety Assays in GxP Environments | Part 1
May 30, 2024
Valentina Armiento
Next-Generation Sequencing (NGS) is a highly sensitive technology that is becoming widely adopted in biopharmaceutical drug development and manufacturing to assess the product characterization and biosafety of biotherapies. Regulatory authorities are establishing guidelines for biopharmaceutical companies on how to use such NGS-based assays, including guidelines for computational systems that support NGS-based assay data analysis within a validated (GxP) environment, to ensure product quality, data integrity, and patient safety. To this end, the International Council for Harmonisation (ICH) adopted the revised Q5A(R2) guideline on viral safety evaluation of biotechnology products which will become effective on June 14, 2024.1 Genedata Selector® is an enterprise software platform that simplifies and accelerates complex NGS-based data analysis workflows and creates a single source of truth across NGS instruments and assays. The software comes with GxP-required functionalities right out of the box. Genedata also offers validation support services to assist companies in their Computerized System Validation (CSV) projects.
This blog post is the first part of a comprehensive two-part series, in which we will examine the regulatory landscape and the requirements associated with the implementation of NGS-based assays in a validated environment.
Unfolding the Potential of NGS-Based Assays
Within biopharma companies, Next-Generation Sequencing (NGS) has become a powerful tool to ensure the quality of biotherapeutics. It provides cost-efficient sequencing of DNA and RNA and can be thus used to detect the identity of genetic variants and mutations and to confirm the integrity of biotherapies and the underlying bioprocess. NGS is expected to be increasingly used in GxP environments, replacing classical non-NGS-based assays, especially for biosafety assessment, where it becomes the game changer. It helps prevent biological contamination like adventitious agents throughout the manufacturing process from starting material to batch release or sterility testing. This ensures the safety and efficacy of products, including gene therapies and vaccines, making NGS an indispensable tool in the field.
Compared to such classical assays, NGS-based assays are a valid alternative in terms of speed, cost-effectiveness, and sensitivity.2 The importance of the applications of NGS was also confirmed during the industry panel discussion of the 8th Genedata Selector NGS Open Forum. A poll conducted with participants from more than 30 biopharmaceutical companies revealed that NGS-based assays are most frequently used by such organizations for adventitious agent detection (AAD) and variant detection. It reflects the industry's commitment to biosafety and quality control.
Improved master cell bank (MCB) release testing is crucial for biotherapeutics and antibody production, thus NGS becomes the method of choice to ensure cell line product quality across sites and batches. Moreover, NGS finds application in monitoring other critical quality attributes (CQA) like identity, integrity, and even potency of cell and gene therapy products. By providing molecular insights into the genetic composition and potential variations, it verifies the absence of undesirable mutations or alterations, thereby upholding the quality and compliance of these products with Good Manufacturing Practice (GMP) regulations.
The Good Manufacturing Practice in Analytical Development
GMP in the biopharmaceutical industry ensures the safety and quality of biotherapeutics for patients. It comprises a set of stringent requirements that drug manufacturers must comply with throughout the entire pharmaceutical production process to minimize risks associated with biopharmaceutical manufacturing, such as unexpected contaminations and mix-ups that could harm patients’ health. Biopharmaceutical companies maintain the integrity and traceability of their products, guaranteeing that every batch of a drug is safe and of the highest quality to fulfill GMP regulations. Thus, GMP plays a significant role in assuring quality and patient safety.
In analytical development, assay validation is one of the critical components of GMP. It is a systematic process that demonstrates the accuracy, reliability, and consistency of an analytical method used to assess the quality and safety of biotherapeutics. The validation process is essential to ensure that the analytical methods used in GMP environments provide trustworthy results. The process of assay validation involves several steps that must be followed: confirming the selectivity, assessing accuracy, specificity, linearity, and precision, and establishing a valid range over which the assay can be conducted.3
The Fast Evolution towards the Acceptance of NGS-Based Testing by the Authorities
Although the validation of NGS-based assays in the GMP environment is still underway, it becomes clear that NGS-based testing is applicable in a regulatory setting with numerous advantages. Moreover, such validation can unlock the full potential of NGS in the pharmaceutical industry. To this end, in 2018 the FDA finalized two guidelines that outline the steps required to design, develop, and validate NGS-based testing.4 The first guideline focuses on accessing and utilizing FDA-recognized databases to support the clinical validity of NGS-based tests.5 The second guideline provides key considerations for designing, developing, and establishing NGS-based tests, which can then streamline the premarket review process and aid in the diagnosis of suspected germline diseases.6 These documents outline a framework for the agency to streamline the development and review of NGS tests. In addition, the International Council for Harmonization (ICH) of Technical Requirements for Pharmaceuticals for Human Use has developed the Q5A guideline. This guideline outlines the testing and evaluation procedure for the viral safety of biological products and specifies the required data to be submitted for product registration. The final ICH Q5A(R2) guideline1, 7 recommends the NGS technology and its use as complementary in vitro cell-based infectivity assays to the in vivo animal testing assays due to its high sensitivity in virus detection, reducing animal use and timelines. However, to deploy NGS in a GMP environment, a validation package of the NGS workflow, from wet lab protocol to lab procedures and data analysis, must be provided to the authorities.1
Biopharmaceutical companies face significant challenges as they enter GMP-regulated environments. The primary challenge is to comply with strict regulatory requirements, including ensuring data integrity and maintaining security while handling sensitive information. In this context, the Computerized System Validation (CSV) of data and NGS workflows presents several aspects. The sequencing instrument comes equipped with specialized software, each requiring a unique validation effort in terms of time and resources. The sequencing technologies and instruments provided by Illumina, Pacific Biosciences, and Oxford Nanopore Technologies contribute to a growing number of modality-specific integrity, identity, potency, and safety assays.
As biopharmaceutical companies move forward to pursue innovation and are eager to use an ever-increasing range of NGS assays, they need to implement a flexible and versatile solution that allows for the validation of multiple NGS assays derived from different methods, instruments, protocols, and facilities.
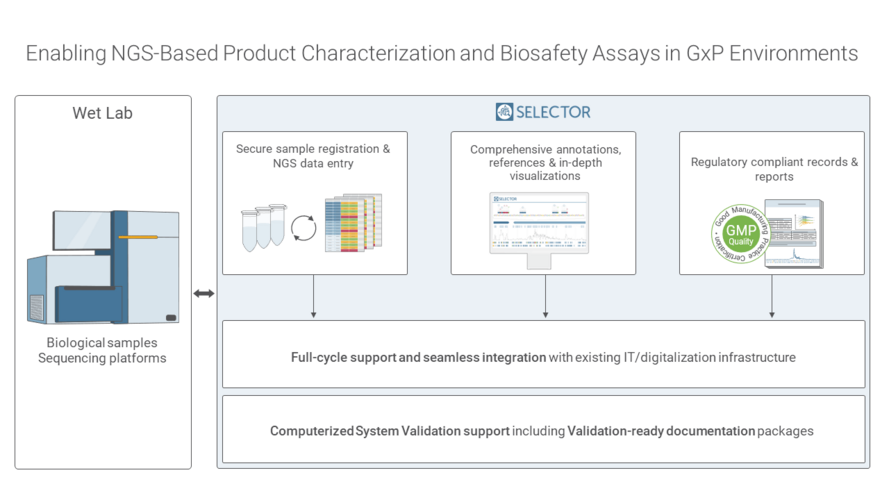
Addressing the Challenges of NGS-Based Assay Validation
To effectively address the end-to-end challenges that arise during the validation of NGS-based assays, companies require a validated platform that acts as a single source of truth and allows tracking of the samples from registration through analysis across different instruments, both within and between sites or organizations.
The Genedata Selector team has extensive expertise and years of experience in developing validation-ready solutions for various Next-Generation Sequencing (NGS) based assays. Collaborating with industry partners, the Genedata team customizes solutions that are not only specific and reliable but also meet the unique requirements of each customer project. Genedata Selector is a software solution that can greatly assist biopharmaceutical companies in navigating the challenges of operating in a GMP environment. The software complies with the FDA's 21 CFR Part 11 regulation, which establishes the basic criteria for validating electronic signatures, provides a platform for managing data while ensuring the authenticity, integrity, and confidentiality of records, and supports customers with a validation-ready documentation package. It streamlines GMP-compliant workflows for NGS-based assays, providing a single source of truth that empowers scientists to integrate, analyze, and share all project-related data for informed decision-making. The platform standardizes and automates sample registration, NGS data processing, and analysis to reporting with easy-to-use "Playbooks". It centralizes all data-related activities and transforms data into insights for more efficient collaboration. With Genedata Selector, biopharmaceutical companies accelerate standardized data processing and analysis with automated workflows, as well as their validation path and submission to regulatory authorities.
Conclusion
NGS is a game changer for biopharmaceutical drug development and manufacturing processes. Its potential and complexity have led regulatory authorities to establish an increasing number of guidelines to validate NGS-based assays and systems within a GMP environment, ensuring the integrity of the data assays and reports. By selecting Genedata Selector, biopharmaceutical companies can simplify the NGS-assay validation process in-house and ensure compliance with quality assurance requirements in a very short time. Genedata Selector experts work closely with customers to validate multiple NGS systems, enabling biopharmaceutical organizations to deliver safe products to patients in a fast manner.
In conclusion, this blog post represents the first part of a comprehensive exploration of the regulatory landscape and the requirements associated with the implementation of NGS-based assays in a validated environment. We encourage you to read part 2 of this series for further insights on the importance of CSV and how to accelerate assay validation with Genedata Selector and our validation support services. In the meantime, if you have any questions, please feel free to contact us at selector@genedata.com or request a demo of our solution.
Request Genedata Selector Demo
References
- International Council for Harmonisation (ICH), European Medicines Agency (EMA). Viral Safety Evaluation Of Biotechnology Products Derived from Cell Lines of Human or Animal Origin Q5A(R2). (2023).
- Müllberg Jürgen. Rapid Methods for Adventitious Virus Detection Acceleration of Clinical Timelines/“Speed to Patient”. American Pharmaceutical Review. (2020).
- Food and Drug Administration (FDA). Process Validation: General Principles and Practices. (2011).
- Luh Frank, Yen Yun. FDA guidance for next generation sequencing-based testing: balancing regulation and innovation in precision medicine. npj Genomic Med 3, 28 (2018).
- Food and Drug Administration (FDA). Use of Public Human Genetic Variant Databases to Support Clinical Validity for Genetic and Genomic-Based In Vitro Diagnostics. (2018).
- Food and Drug Administration (FDA). Considerations for Design, Development, and Analytical Validation of Next Generation Sequencing (NGS) - Based In Vitro Diagnostics (IVDs) Intended to Aid in the Diagnosis of Suspected Germline Diseases. (2018).
- International Council for Harmonisation (ICH), European Medicines Agency (EMA). Press Release: ICH Assembly Meeting, Prague, Czech Republic, October/November 2023. (2023).