NGS Revolution for Cell and Gene Therapy Products: Establishment & Monitoring of Critical Quality Attributes
January 30, 2024
Marie-Ange Kouassi
The quest to make life-changing cell and gene therapies (CGTs) more accessible is currently impeded by the slow adoption of innovative approaches that ensure timely quality and safety. An examination of the critical quality attributes of CGT products reveals that replacing current assays with a comprehensive NGS assay would not only preserve biological material and decrease costs but also accelerate investigational new drug (IND) applications and ultimately treatment availability.
The potential to reverse currently incurable diseases with cell and gene therapies is immense. However, ensuring their safety is a major challenge that limits their widespread use. While some cell and gene therapies like Tecartus and Luxturna have shown remarkable outcomes1, there have been instances where other therapeutics have led to life-threatening unforeseen outcomes and clinical holds.2,3 A likely cause? The companies developing these treatments are under pressure to deliver safe biological material under tight timelines. To prevent unforeseen results and ensure the development of the intended therapeutic, in-process testing is required to ensure these therapeutics uphold their critical quality attributes (CQAs). Regulatory authorities such as the FDA and the EMA require comprehensive documentation about CQAs when companies submit an IND application. Establishing and monitoring these attributes throughout development and manufacturing can help to ensure the product developed is safe and fit for its purpose. For this, a streamlined assay portfolio and the right technology that allows determining multiple attributes of a therapeutic simultaneously would be highly beneficial.
What Are CQAs and How Do They Differ for Cell or Gene Therapies?
A Critical Quality Attribute has been defined as a physical, chemical, biological, or microbiological property or characteristic that should be within a suitable limit, range, or distribution to ensure the desired product quality.4
When it comes to cell therapies, which are likely prone to impact by the origin of the starting material (patient/donor characteristics such as age, genetic makeup, etc.) and their external environment, CQAs include identity, potency, and safety.
- Identity: the product contains the intended cellular components
- Potency: the product has the biological function relevant to treating the clinical indication
- Safety: the product is free from viable adventitious agents (e.g., mycoplasma, bacteria, viruses) and foreign matter (e.g., endotoxins, proteins, vectors, DNA, unwanted cells, or culture reagents )
Safety can be further separated into two aspects: Sterility, which focuses only on contaminating microorganisms, and Purity which refers to process impurities. The type of cell therapy (e.g., pluripotent, multipotent, and unipotent stem cells) determines the specific assays used to establish CQAs. However, the increasing number and variety of cell therapies leads to a lack of standardization between organizations.5 This variation makes it even more essential to perform in-process testing and characterization but presents challenges when it comes to ensuring consistency and assessment by regulatory authorities.
For gene therapies, the genetic material is packaged within a vector delivery system (e.g., viruses), therefore the identity is known. Nonetheless, it is equally crucial to verify the integrity, potency, and safety of the product. According to the FDA, the method used to verify potency in vivo must correspond to the biological properties of the product.5 This includes the ability of the vector to transfer the gene and the biological effect of the expressed genetic sequence.
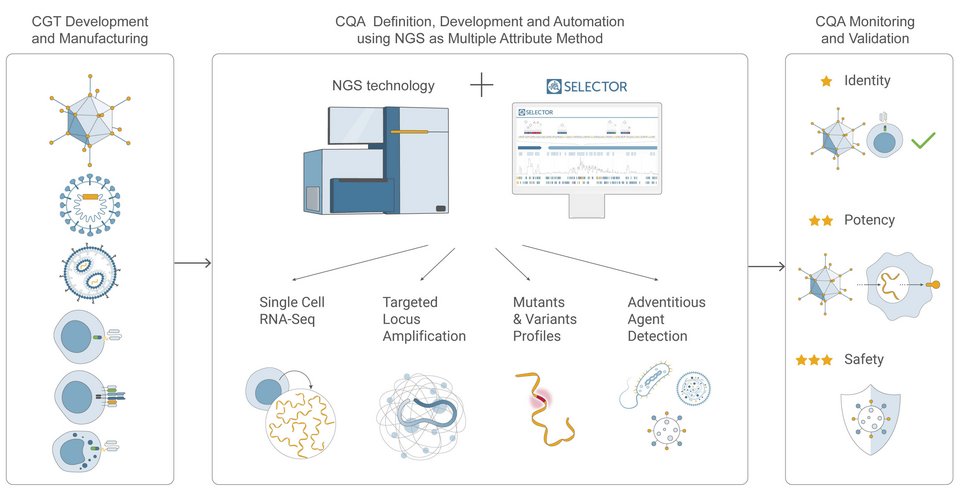
How Can NGS Help to Monitor CQAs of CGT?
Next-Generation Sequencing (NGS) is an advanced technique that enables comprehensive characterization of biotherapeutics using a range of innovative approaches. For example, single-cell RNA-Seq, an NGS-based approach, allows the examination of heterogeneity in a sample of cells – this can be extremely valuable in confirming the identity, potency, and purity of cell and gene therapy products.. NGS is also beneficial for biosafety testing by detecting the presence of adventitious agents, thereby allowing safety confirmation. Importantly, NGS can also help in the early detection of potential issues or anomalies during the CGT development process, such as unexpected mutations or changes in gene expression, which could impact the safety or efficacy of the therapy. The NGS-based approach known as targeted locus amplification (TLA), a Cergentis-developed technology, allows for verification of gene integration sites, determination of the copy number of genes inserted into cell therapy products, or analysis of the potential for insertional mutagenesis, and many more. In short, NGS is now the multi-attribute method for CGT products as it eliminates the need for implementing several different assays to determine CQAs by streamlining multiple assays. The vast amount of useful information derived from NGS simplifies, accelerates, and makes it more cost-efficient to answer the questions:
- Is my product what I think it is? (Identity)
- Is my product able to fulfill its intended purpose? (Potency)
- Is my product contaminated with other organisms? (Safety)
Furthermore, NGS is gaining increasing interest as a commonly used technique for CQA establishment and monitoring. It is not surprising that the European Medicines Agency requested the developers of Zolgensma, the gene therapy for spinal muscular atrophy, to perform NGS to assure vector genome integrity.6
However, handling NGS data can be intricate if a company lacks the resources to automate the processing and generation of insights from the vast amount of data produced. Without this, data-informed Go/No-Go decision-making is delayed during development and manufacturing.
Overcoming the Complexity of NGS with Genedata Selector®
Genedata Selector empowers biopharma companies to accelerate the development of safe cell and gene therapy products by accurately confirming identity, potency, sterility, and purity. With the help of Playbooks, this enterprise software platform simplifies and automates NGS data analysis for users, regardless of their bioinformatics experience level. This allows leveraging the comprehensive insights that can be obtained from NGS-based assays throughout the CGT development and manufacturing process. For cell therapy, Genedata Selector automates the analysis and visualization of data from scRNA-sequencing, CRISPR events, and TLA analysis, to confirm identity and monitor cell characteristics after genetic manipulation. For gene therapy, besides allowing safety confirmation through adventitious agent detection (e.g., viral contamination, wrongly packed nucleic acids), the software aids the generation of mutation profiles to assess potency. Designed to streamline submission to regulatory authorities, this future-proof platform tracks all sample information, including genomic and phenotypic information, and generates automated detailed reports.
While there has been significant focus on CGT development technologies and manufacturing processes, innovative methods for maintaining in-process critical quality attributes are still lacking. Also, the wide diversity of therapeutics has led to discrepancies between developing organizations and an increasing number of assays being used to establish CQAs. By harnessing the full power of NGS using Genedata Selector biopharma companies can significantly reduce the number of assays from 20 to approximately 4, allowing for standardization and automation for GMP-compliant in-house processes. The software not only simplifies the data management, processing, analysis, and visualization but also the conversion of data into actionable results that support Go/No-Go decisions from assay definition and development to standardization and automation. By choosing Genedata Selector, companies can ensure well-documented, transparent, and seamless cross-collaboration with internal and external partners while benefitting from interactive communication tools all within a single point of truth. This facilitates efficient collaborative confirmation of CQA products that serve their intended purpose while ensuring patient safety.
References
- Insights on Successful Gene Therapy Manufacturing and Commercialization. Cell Culture Dish, (2020).
- Angus Liu. 2 deaths After Novartis’ Zolgensma Put Gene Therapy’s Liver Safety in the Spotlight Once Again. Fierce Pharma (2022).
- Jane Byrne. Deaths Trigger Pause in Ceylad’s Allogeneic CAR-T Trial. BioPharma-Reporter.com (2022).
- Committee for Human Medicinal Products, European Medicines Agency (EMA). ICH Guideline Q8 (R2) on Pharmaceutical Development Step 5 (2017).
- Francis Karanu et al. Improved Harmonization of Critical Characterization Assays Across Cell Therapies. Regenerative Medicine (2020).
- Committee for Medicinal Products for Human Use (CHMP) and Committee for Advanced Therapies (CAT), European Medicines Agency (EMA) Zolgensma Assessment Report (2020).