Automating NGS-Based Vector Integrity Confirmation to Ensure Quality Gene Therapy Products
April 27, 2023
Marie-Ange Kouassi, Qiu Ruan, Marcell Veidner, Devon Ryan
Using gene therapies, it is now possible to treat previously incurable inherited diseases. Yet, manufacturing these complex biological therapies to reach the necessary quality and safety standards for clinical success is challenging. With Genedata Selector®, biopharma companies are empowered to reap the benefits of NGS by embracing quality by design to accelerate the development of safe and effective therapies.
What an exciting time! Gene therapy development is occurring at warp speed thanks to the in-depth knowledge gathered on viral vectors over the years. Adeno-associated viruses (AAVs) have proven to be the most promising for administering cell function-altering genetic material to patients suffering from life-threatening diseases. This is due to their versatility, low level of immunogenicity, and low vector-related toxicity compared to adenoviruses (Ads) and lentiviruses (LVs).1 Many biopharma companies are thus developing AAV-based gene therapies- with several approved drugs already on the market (e.g. Glybera, Luxturna, and Zolgensma).2 Yet, during manufacturing, there are challenges due to heterogeneity as well as contamination with product- and process-related impurities which ultimately reduce their quality, safety, and efficacy.3 Ensuring viral vectors have the right identity to bring about the synthesis of the right protein and the desired therapeutic effect, is crucial. This is why performing variant detection is of utmost importance. Next-Generation Sequencing (NGS) is a powerful unbiased multi-attribute method (MAM) that enables this critical quality attribute assessment as well as the detection of unknown contaminants. However, leveraging NGS successfully during analytical development requires bioinformatics expertise to generate insights that support better decision-making and reporting. Genedata Selector® is a sequencing technology-agnostic enterprise software that facilitates data-informed decision-making during gene therapy development by empowering the various stakeholders involved. The platform simplifies what presently is a complex process providing teams with accuracy, traceability, and support for GMP validation, as well as greater efficiency throughout cross-functional collaboration for gene therapy development.
The production of recombinant AAVs (rAAVs) involves several important steps; the construction of the transfer plasmid from the AAV vector as well as the transgene and finally, the packaging of the rAAV genome into viral particles.4 The entire rAAV genome is a recombinant single-stranded DNA comprising a transgene and expression-regulating events flanked by two inverted terminal repeats (ITR) sequences. During the rAAV DNA production process of gene therapy development, common mutations (e.g. substitutions, insertions, duplications, and deletions) can occur anywhere in the rAAV DNA sequence, including the transgene and ITR regions. Mutations within the transgene can lead to the synthesis of a non-functional protein that fails to facilitate the desired therapeutic effect, or altered protein levels while mutations in the ITR can lead to packaging failures.5 It is, therefore, crucial to have a sensitive assay in place to perform sequence identity and integrity testing of the transfer plasmid as well as the packaged rAAV DNA. Detecting and tackling these mutations as soon as possible helps to ensure a successful production cycle and mitigates the risk of losing time and valuable resources due to the development of an inadequate gene therapy product that does not meet the required safety and quality standards. Mutations within the transgene can occur at a very low frequency, thus, the sequence identity testing assay needs to rely on a highly sensitive method. Compared to traditional Sanger sequencing, NGS-based sequencing methods offer much greater sensitivity facilitating the identification of low-frequency mutation events, even as low as 1%. To analyze the data generated from NGS for data-informed decision-making during gene therapy development, a bioinformatics pipeline is required that aligns sequencing reads to a reference sequence and then detects mutations using a variant calling algorithm. Not only can the bioinformatic analysis pipeline be set up to detect mutations but it can also predict the potential effects of these mutations. By assessing the frequency and the potential effect of a given mutation, the various stakeholders within an analytical/process development team can be equipped with the necessary metrics to make the right go/no-go decisions during gene therapy development. Throughout this process, it is important to ensure all stakeholders have access to the right product-related data, process-related data, and resulting insights (linked to the samples they originated from) to be able to successfully collaborate and quality control their gene therapy products.
Within biopharma companies, Bioinformaticians play a key role in NGS-based quality control of gene therapies. They essentially develop methods for data processing and analysis so the results can be interpreted by Analytical Scientists to detect deviations in quality, safety, and efficacy amongst samples. Typically, this involves building NGS data processing pipelines to progress with their own work and analysis pipelines that their team members can use. A challenge when it comes to NGS data is ensuring standardization in data processing and analysis. Although Bioinformaticians are not directly involved in regulatory submissions, they need to ensure everything they do is tracked, and that the data lineage is maintained for other team members providing transparency throughout the process. Genedata Selector gives full control to Bioinformaticians allowing the standardization of data processing and analysis by easily building workflows within the platform. Designed with built-in logic for automated NGS analyses, Genedata Selector is also equipped with out-of-the-box workflows that users can leverage to quickly get started – these are configurable to user experiments, data types, and samples. Once Bioinformaticians are satisfied with the performance and outputs of their designed workflows, they can “lock” them and convert them into guided user interfaces named “Playbooks” for their team members to use. These Playbooks can be run simply by selecting the data source, genomics references, and viral database. They guide users through a variety of analyses, such as mutation profiling (for variant analysis), adventitious agent detection, and plasmid contamination checks. Rather than using multiple different tools, reducing efficiency, Bioinformaticians are equipped with a single unified interface in the Genedata Selector platform where they can intuitively keep track of all their workflows and efficiently communicate with team members regarding data activities.
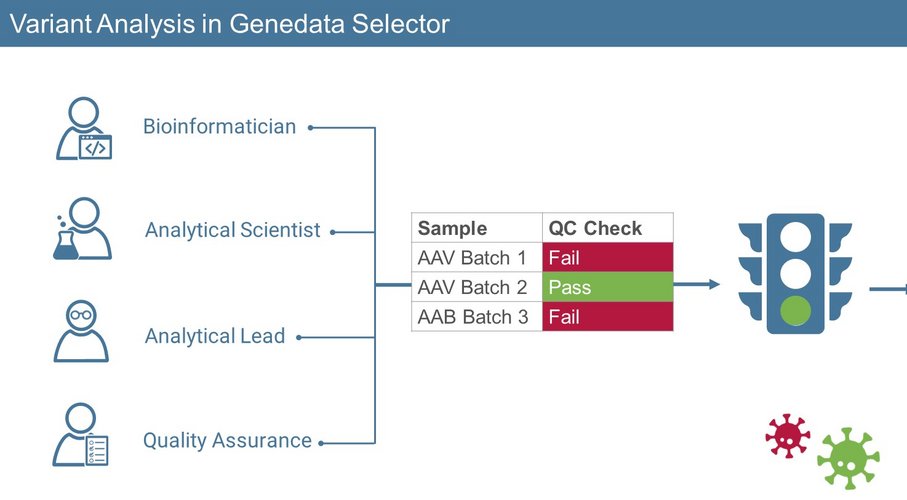
As the operators of NGS-based assays during gene therapy development, Analytical Scientists are in pursuit of clear, actionable insights regarding which samples are likely to be successful in production or not. After running their experiments, they analyze their resulting data to identify which samples should progress in the development and manufacturing process. This involves inspecting for any impurities that could impact vector efficacy, safety, and quality, for example:
- Does the vector contain the transgene of interest?
- Are there any variants or truncations of the vector?
- Does the capsid contain any impurities (e.g. nucleic acids)
- Are there any adventitious agents present?
Often, the progress of Analytical Scientists can be delayed as they may not be able to immediately generate insights from their data without support from bioinformatician experts. With Genedata Selector, Analytical Scientists gain independence as they leverage the Playbooks- workflows pre-built and approved by their bioinformatician team members. These workflows exist as easy-to-use, point-and-click guides that automate complex NGS Analyses for efficient result generation. In Genedata Selector, the results are displayed using a unique traffic light system that clearly indicates which samples have met the required standards or not. These go/no-go signals governed by previously set thresholds are key as they significantly accelerate decision-making. If a sample fails during variant detection, scientists can investigate the result in detail using the genome viewer provided in Genedata Selector. This allows them to dive deeper on a genome and sequence read level to identify the location and type of mutations detected. With this, the interpretation of results and troubleshooting is simplified and accelerated. All experiments run in the Playbook are tracked in timestamped reports which contain the full chain of custody documenting all data-related activities of users. The results are also linked to samples and any associated metadata. This makes it easier to report insights to the Analytical Lead or to provide the Quality Assurance team with documentation for submission to regulatory authorities.
Throughout the gene therapy development process, the Analytical Lead requires a global overview of the different samples being analyzed and the results generated from them. They need to be able to quickly gain insights regarding critical quality attributes as well as understand and assess the various processes undertaken. Monitoring processes would enable them to identify whether a methodological change or a different technological solution is required for process optimization. With Genedata Selector’s comprehensive overview of results, Analytical Leads can always “be in the know”, staying aware and well-informed of the progress of samples throughout the development cycle, without needing to spend excessive hours navigating through uninformative, cryptic reports.
Quality is central to the development of gene therapies and the main priority of Quality Assurance (QA) Experts along with GxP validation. To ensure that the best standards are met for the end product, QA experts oversee the entire process to confirm compliance with the regulatory requirements. From sample registration, and wet lab experimentation to sequencing, data processing, analysis, and reporting, Quality Assurance Experts develop standard operating procedures (SOPs) and aggregate documentation regarding the entire assay. The QA experts also ensure their teams are following protocols that meet the requirements of a GxP-validated environment. This involves having the necessary documentation, which contains the full chain of custody and provides evidence that all computerized systems are suitable and performant for the use cases involved in the development process. In Genedata Selector, there is a complete audit trail within the reports, as well as built-in permission controls governing who can run certain analyses. Moreover, these reports contain indications about the software version. The fields of these reports are customizable in case regulators require specific types of information. Automating the process with Genedata Selector eases the burden of audits, as all the necessary information is located in a central place for easy access during regulatory submissions.
Gene therapy development is an intricate process because of the nature of the various components of viral-vector-based therapies. Changes to the product or process may hinder the therapy from being able to bring about the intended outcome or could result in adverse effects. This is why it is essential to perform variant detection and define therapy-specific success criteria up front for monitoring quality throughout the development cycle. NGS alleviates the need for biopharma and biotech companies to perform multiple different assays while Genedata Selector makes insight generation from NGS data simple by empowering every individual in gene therapy development with the necessary tools to do their best work. With Genedata Selector, users can generate comprehensive results regarding different critical quality attributes of gene therapy products. Besides the software, our scientific consultants are available to provide you with support for assay definition, building workflows, and bioinformatics expertise. The dedicated IT team of Genedata ensures seamless integration of the software into your infrastructure and with any internal legacy systems. For any support regarding validation, you have access to our QA team who have a wealth of experience with GMP and can provide templates to support your performance qualification (PQ). To learn more about Genedata Selector and its support for variant detection during gene therapy development, contact us for a demo.
Marie-Ange Kouassi, Ph.D., Scientific Marketing Specialist, Genedata AG
Qiu Ruan, Ph.D., Scientific Consultant, Genedata Inc.
Devon Ryan, Ph.D., Head of Scientific Consulting, Genedata AG
Marcell Veidner, Business Development Manager, Genedata AG
References
1.Zaiss AK et al. Differential activation of innate immune responses by adenovirus and adeno-associated virus vectors. Journal of Virology (2002).
2. Bulcha, J.T. et al. Viral vector platforms within the gene therapy landscape. Signal Transduction Targeted Therapy 6, 53 (2021).
3. Fuentes C et al. Beyond empty and full: understanding heterogeneity in rAAV products and impurities. Dark Horse Consulting Group Whitepaper (2023).
4. Nature Portfolio. Three is the magic number in gene therapy production. Accessed Online on 27.04.2023.
5. Tam Tran N et al. AAV-Genome Population Sequencing of Vectors Packaging CRISPR Components Reveals Design-Influenced Heterogeneity. Mol Ther Methods Clin Dev (2020).