Broad Institute Scientists Search for Treatments for COVID-19 Using High Content Screen
July 28, 2020
Ada Yee
As the COVID-19 virus continues to sicken people worldwide, researchers everywhere are racing to find candidate drugs to treat this new illness. High-content screening (HCS) and high-throughput screening (HTS) are key tools in this effort. Through one such HCS screen, scientists at the Broad Institute have reported in a recent preprint a possible candidate for fighting the devastating respiratory consequences of COVID-19.1
Mucin-1 (MUC1) is a protein found on the membrane of mucus-secreting epithelial cells, including those that line the airway. MUC1 is upregulated in association with lung injury and pulmonary complications. Given this role of MUC1 and the acute respiratory distress characteristic of COVID-19, researchers Alimova et al. reasoned that MUC1 could be an interesting target for treating COVID-19.
Recently, these scientists had conducted a screen for compounds to treat a rare kidney disease involving mutant MUC1.2 These results also contained data on wildtype MUC1, so they were immediately able to go back and mine the data for hits that reduce MUC1. This screen included 3,713 compounds from the Broad Repurposing Library—all of which are already being investigated at the preclinical and clinical levels—and each compound was tested in a 5-point dose response series. This was followed by a secondary screen on 203 compounds at 10 doses each. Data were collected on the Opera PhenixTM High Content Screening System (PerkinElmer) and analyzed in Genedata Screener®.
The authors wanted a compound that (1) functions by reducing protein instead of mRNA transcript, because the latter has not proven successful in the clinic, and (2) is not toxic to cells. Using qPCR and cell viability screens, they finally homed in on four such candidates. They went on to test the orally administrable version of one of these, fostamatinib, in an in vivo model of acute lung injury and showed it reduces MUC1. Fostamatinib is already FDA-approved for use in other indications, meaning that it could potentially pass through development and safety tests and reach COVID-19 patients much sooner than and entirely novel drug.
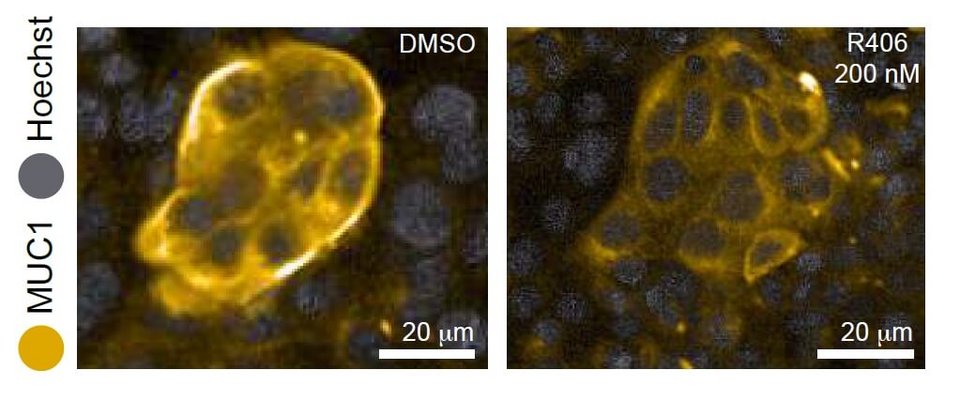
Though it remains to be seen whether fostamatinib can alleviate symptoms in animal models of COVID-19 infection, these early results (the paper has not yet been peer-reviewed) are promising. Certain cytokines observed to be increased in COVID-19 patients as part of a “cytokine storm”3,4 appear in respiratory syndromes where MUC1 plays a role and have been shown to regulate MUC1.5 Furthermore, early evidence suggests that in patients with the most severe COVID-19 cases—those requiring ventilators and intubation—MUC1 is elevated.6 Above all, this work is an excellent example of how high content screens and repurposing libraries are being used to speed up the fight against the current pandemic.
Learn more about Genedata Screener for HCS and our integrations with PerkinElmer instruments and other imaging systems.
Author: Ada Yee, Ph.D., Scientific Story Researcher, Genedata Screener.
References:
1. Alimova et al. A High Content Screen for Mucin-1-Reducing Compounds Identifies Fostamatinib as a Candidate for Rapid Repurposing for Acute Lung Injury during the COVID-19 pandemic. BioArXiv [Preprint]. June 30 2020 [cited 2020 July 22].
Update October 29, 2020: A peer-reviewed version is now published in Cell Reports Medicine 1:8 100137 (2020).
2. Dvela-Levitt et al. Small Molecule Targets TMED9 and Promotes Lysosomal Degradation to Reverse Proteinopathy. Cell 178:3 521-535 (2019).
3. Moore JB and June CH. Cytokine release syndrome in severe COVID-19. Science 368:6490 473-474 (2020).
4. Aziz et al. Elevated interleukin‐6 and severe COVID‐19: A meta‐analysis. Journal of Medical Virology 1-3 (2020).
5. Li et al. Macrophage-derived interleukin-6 up-regulates MUC1, but down-regulates MUC2 expression in the human colon cancer HT-29 cell line. Cell Immunol 256:1-2 19–26 (2009).
6. D’Alessandro et al. Serum KL‐6 concentrations as a novel biomarker of severe COVID‐19. Journal of Medical Virology 1-5 (2020).