Faster, Safer, More Comprehensive Viral Detection in Biologics Manufacturing
October 6, 2022
Marie-Ange Kouassi
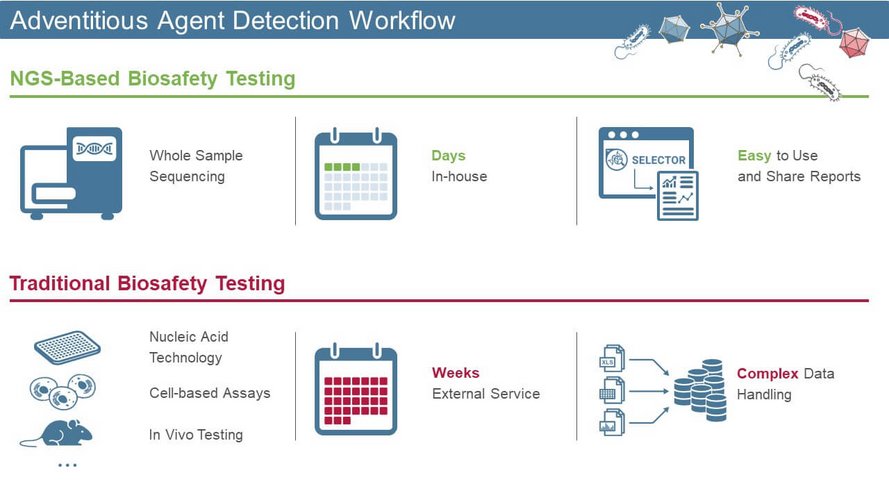
Failure to detect adventitious agents early during biologics production can lead to facility downtime and severe economic losses. Traditional biosafety testing takes approximately 4–5 weeks, which severely delays subsequent manufacturing stages. However, Next-Generation Sequencing (NGS) is an attractive alternative to existing methods as it allows highly sensitive detection of adventitious agents within only a few days. When applied throughout the biologics manufacturing process, NGS can significantly accelerate clinical timelines.
In modern healthcare, the most advanced therapies for treating disease are derived from or produced by living cells. Commonly referred to as biologics, examples include proteins such as monoclonal antibodies and insulin as well as, fragments of DNA or RNA delivered via viral vectors, and vaccines. The manufacturing of biologics presents unique challenges, not only in their complexity, but also in their biosafety requirements.
Biotherapeutics, including new cell and gene therapies are vulnerable to contamination (e.g., by viruses). These contaminants must be detected early to prevent delays in the production process which would impact the time to market of the therapy. Dr. Jürgen Mülberg, a viral biosafety expert at Bristol Myers Squibb, highlighted Next Generation Sequencing (NGS) as a promising alternative to existing testing methods as it has the benefit of yielding significantly faster results.1
Traditional viral safety test methods include in vivo testing, indicator cell-based infectivity assays, and nucleic acid technology (NAT)-based tests. In vivo animal models are challenging as they take approximately 4–5 weeks delaying the subsequent manufacturing stages, downstream decision-making, and clinical stages. They have also been shown to be less sensitive and reliable than alternative molecular methods such as PCR. Although more reliable, PCR-based tests are also limited as they cannot detect unknown viruses in viral biopharmaceuticals. NGS enables unbiased testing to detect known and unknown viruses, while providing results more quickly.
A targeted NGS method can be used by manufacturers to avoid contamination of entire facilities and production batches through early, highly sensitive detection of adventitious agents.2
Regulations outlined in the Code of Federal Regulations (CFR, Title 21), the US, and European Pharmacopeia, state that while manufacturing biologics, viral safety measures are crucial.3 Such measures exist in three pillars – Prevention, Detection, and Removal – and consider aspects of manufacturing facility design, control techniques, testing stages, and purification processes to remove impurities. Currently, regulatory bodies such as the WHO, FDA, and the European Pharmacopeia Commission, recognize the advantages NGS brings and encourage substituting in vivo testing.3,4,5
Failure to detect adventitious viruses in the production process can lead to facility downtime and severe economic losses. Ultimately, these consequences damage the reputation of a biopharmaceutical company. An example is the minute virus of mice (MVM) that infects rodents and their derivative cells, such as Chinese hamster ovary (CHO) cells. This environmentally stable virus has been problematic for several biopharmaceutical companies6 as early as the 1990s, leading to product losses and delays of treatment delivery to patients.7
NGS has the potential to accelerate clinical timelines providing a faster method of detecting contaminants throughout the biologics manufacturing process. Scientists can use NGS (a targeted assay) to shorten in-process validation times, accelerating the drug development process. With the growing number of novel biologics and biosimilars entering the market, manufacturers could reduce both time and risk by incorporating NGS-based testing into their workflows. However, an advanced computational platform is required to enable manufacturers to efficiently manage the wealth of data generated from NGS for conversion into actionable insights.
Genedata Selector®, the advanced digital platform, and scientific consulting services, offers its support to biopharmaceutical organizations interested in implementing NGS-based biosafety testing in-house. The platform supports the entire biosafety testing process from sample preparation to results management and reporting.
As an intuitive enterprise software solution, Genedata Selector enables the tracking, processing, analysis, and visualization of NGS data in-house eliminating the need for sending sensitive IP to external service providers. As a result, scientists maintain full control of the entire analysis process, without the need for extensive bioinformatics training.
Genedata Selector enables standardization and automation of the analytical process and enables efficient go/no-go decisions on biosafety, including adventitious agent detection (AAD), verification of gene insertion sites, and assessment of cell line stability and product integrity. As a validation-ready platform, Genedata Selector is suitable for deployment in a GxP environment. It is also designed to accurately handle reporting, auditing, and security requirements to comply with regulatory guidelines. Ultimately, the platform helps to maximize resources and accelerate the delivery of treatments to patients with critical health needs.
References:
- Müllberg,J. Rapid Methods for Adventitious Virus Detection. Acceleration of Clinical Timelines/"Speed to Patient". Microbiology, (2020).
- Charlebois, R.L., et al. Sensitivity and breadth of detection of high-throughput sequencing for adventitious virus detection. npj Vaccines 5, 61 (2020).
- Guidance for Industry. Characterization and qualification of cell substrates and other biological materials used in the production of viral vaccines for infectious disease indications. FDA, Center for Biologics Evaluation and Research (2010).
- Recommendations to Assure the Quality, Safety and Efficacy of Live and Attenuated Yellow Fever Vaccines. World Health Organization. Page 12 (2010).
- European Pharmacopeia: Chapter 5.2.3. Cell substrates for the production of vaccines for human use (2014).
- Garnick RL. Experience with viral contamination in cell culture. Dev Biol Stand. 1996; 88:49-56.
- Barone, P.W., Wiebe, M.E., Leung, J.C. et al. Viral contamination in biologic manufacture and implications for emerging therapies. Nature Biotechnology 38, 563–572 (2020).
