Cell Therapy: The Potential for Effective Precision Medicine in Oncology and Beyond
June 7, 2023
Nicholas S. Geraci & Justyna Lisowska
Cell therapy is a transformative, rapidly expanding field revolutionizing the treatment of life-threatening diseases. Conditions amenable to cell therapy include those with no currently available effective treatment, those that are resistant to or fail to improve with conventional therapies, or diseases where current therapeutic strategies require long-term administration of drugs with deleterious side effects and invasive procedures. While cell therapies have proven effective primarily in the treatment of hematological cancers, they can be successfully applied to a broad range of diseases such as neurodegenerative or immune-mediated inflammatory diseases. This article describes some cell therapy approaches used in oncology and highlights developments in other therapeutic areas.
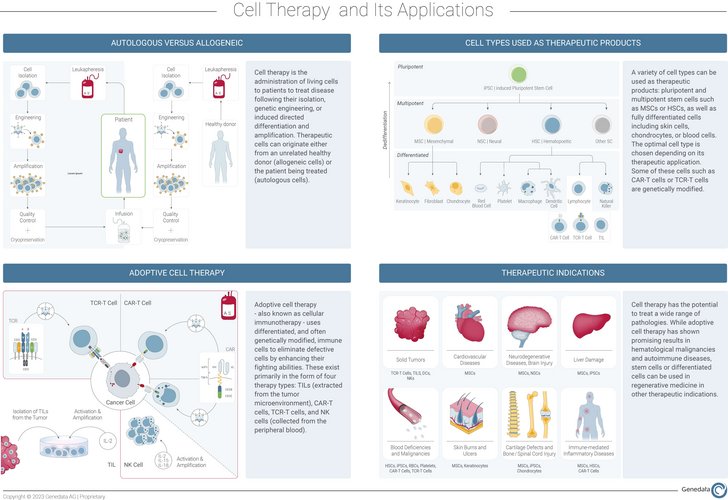
Broadly, cell therapy is the administration of living cells into a patient to treat disease either by eliminating defective cells or regenerating impaired tissues. These therapeutic cells can originate from the patient (autologous) or a donor (allogeneic) and can be of various types depending on the therapeutic application.
Cell therapy development may not only involve the harvesting and reintroduction of the desired cell types to a patient but also require some form of in vitro manipulation such as genetic engineering of selected cells or stem cell differentiation to generate the right cellular phenotype. Typically, desired phenotypes include the ability to specifically recognize and destroy target cells or expand into a particular cellular lineage and replenish impaired tissue with healthy cells. Cell therapies often overlap with gene therapies, which involve the introduction, removal, or alteration of genetic material to modify the expression of a key protein or to produce modified or novel proteins (e.g., using CRISPR methods to generate chimeric proteins).
Current Cell Therapy Approaches in Oncology
In addition to rare diseases, oncology remains the predominant therapeutic indication in cell therapy development.1 This is due to the pressing need to develop solutions for patients suffering from cancers exhibiting refractory or chronic relapsing behavior where traditional therapies do not offer any long-lasting remission. Such patients urgently require efficacious, fast-acting, and precise treatment options that are tailored to their unique clinical presentations and other factors affecting pathophysiology (e.g., genotypes). Cell therapy technologies have the potential to offer this targeted treatment with high degrees of personalization.
Adoptive cell therapy (ACT)—also known as cellular immunotherapy—leverages the natural ability of immune cells to recognize and fight cancer cells. They can be deployed in the form of four therapy types: Tumor-Infiltrating Lymphocyte (TIL), engineered T Cell Receptor (TCR), Chimeric Antigen Receptor (CAR) T cell, and Natural Killer (NK) cell therapies.2 As outlined below, each cell therapy approach carries with it certain advantages for application in the treatment of various cancer types, but also specific challenges.
TIL Cell Therapy
Tumors are often infiltrated by T cells, but these naturally occurring cells may fail to control tumor growth. Before they can effectively kill cancer cells, they must become activated and maintain that state for a sufficiently long time to sustain an effective anti-tumor response. TIL therapy development involves harvesting tumor-infiltrating T cells from the tumor microenvironment (TME), activating, and expanding them in vitro using the cytokine IL-2, and subsequently infusing them back into the same patient. Multiple studies indicate that tumor regression following TIL transfer is mediated by T cells targeting tumor-specific antigens, also known as neoantigens.3 Therefore, enrichment of the final product in neoantigen-reactive T cells is crucial to enhance its clinical efficacy. TILs have been shown as promising in controlling melanomas and this year, we may finally see the first therapy of this type being approved by the FDA: Lifileucel from Iovance Therapeutics is currently being evaluated for the treatment of patients with advanced unresectable or metastatic melanoma.4
TCR Cell Therapy
Not all patients have T cells capable of recognizing and fighting their tumors. These patients need their T cells to be equipped with an enhanced modified T cell receptor (TCR) that enables them to target tumor-specific antigens. TCR-based ACT therapy development involves isolating peripheral blood T lymphocytes, identifying the patient-specific neoantigen repertoire, and genetically engineering the extracted cells to express receptors capable of targeting these neoepitopes with high avidity.5 Yet, TCR activation requires co-stimulation of the accessory signaling molecules and presentation of the targeted antigen via the MHC-I complexes on those target cells, which are often downregulated in cancer.6 Despite these limitations, exciting results from clinical studies are now emerging confirming the efficacy of this strategy in both blood cancers and solid tumors such as melanoma or sarcoma. FDA approval of the first autologous TCR cell therapy for advanced synovial sarcoma, Afami-cel (Adaptimmune Therapeutics), is expected by the end of this year.4
CAR-T Cell Therapy
The limitations of target antigen presentation through MHC-I complexes that plague TIL and engineered TCR therapeutics can be overcome with the use of CAR-T cells. To develop this therapy, peripheral T lymphocytes are extracted and genetically modified in vitro to express the chimeric antigen receptor (CAR) able to specifically recognize and bind natively expressed surface antigens without MHC-I presentation. While this narrows the range of potential antigenic targets, the independence and engineered specificity of those cells provide greater productization opportunities with high degrees of customization. CAR-T cell therapy has demonstrated its therapeutic efficacy in blood malignancies such as acute lymphoblastic leukemia, B cell lymphoma, or multiple myeloma7 giving rise to seven marketed products in the US, Europe, and China. These include Kymriah (Novartis), Yescarta (Kite Pharma/Gilead), Tercatus (Kite Pharma/Gilead), Breyanzi (Celgene/BMS), Abecma (bluebird bio), Carvykti (Legend Biotech) and Carteyva (JW Therapeutics).1
Instead, treating solid tumors with CAR-T cells is challenging. Clinical trials show that although CAR-T cells can engraft in the peripheral blood, traffic to tumors, and respond to target antigens, they fail to expand, persist, and mediate tumoral expansion or induce regression.8,9 The solid tumor microenvironment (TME) entails immunosuppressive cells (e.g., regulatory T cells) and molecules (e.g., PD-L1, IL-10, TGF-β), hypoxia, and nutrient shortages which must be overcome by CAR-T cells to thrive and fight the tumor. Better characterization and understanding of this heterogeneous, complex immunosuppressive environment will be key to developing efficacious CAR-T cell therapies.
While all currently approved CAR-T cell therapies are autologous, efforts are continuously being made to generate off-the-shelf cellular products for use in multiple patients. Companies like Century Therapeutics leverage the unlimited self-renewing capacity of induced pluripotent stem cells (iPSC) and genetic engineering to develop a wide range of fit-for-purpose allogeneic therapies including CAR-T or CAR-NK cells for the treatment of both hematological and solid tumor malignancies.
NK Cell Therapy
NK cells are different from T cell-based therapies in that they express a repertoire of inhibitory and activatory surface receptors that are germline-encoded, and therefore they do not require recombination and clonal selection. This enables them to recognize and rapidly act—by releasing cytotoxic granules to directly lyse malignant tumor cells—without prior sensitization. NK cells are not only stronger candidates for allogeneic product development but may also be genetically engineered for precision targeting (i.e., CAR-NK cells). As with all ACTs, there are many challenges to overcome in NK cell therapy development, such as the difficulty to meet clinical-grade ex vivo expansion, limited in vivo persistence, infiltration into solid tumors, and tumor editing to evade NK cell activity.10
All ACT approaches carry the risks of low cellular expansion, cellular dysfunction, loss of function due to tumor heterogeneity, antigen loss (the time from cell extraction to reinduction must be narrow to ensure the capture of an effective window for treatment), and toxicity. Yet, the promise of these therapies to combat elusive cancers is extremely alluring and has already shown some promise in the market.
Finally, innate immune system cells—including dendritic cells and macrophages— found in the TME can modulate the adaptive immune response to fight cancer.11 They are now being explored for their therapeutic potential as they reveal promising effects, especially for solid tumors against which T-cell therapy has limited efficacy.12
Delivering Cell Therapies to Other Therapeutic Areas
While the use of immune cell therapies has found a niche in the realm of oncology, highly promising early clinical results have been recently reported for autoimmune diseases. A study published in September 2022 demonstrated that a single dose of engineered anti-CD19 CAR-T cells administered to five systemic lupus erythematosus (SLE) patients led to the depletion of their B cells, the main source of pathogenic antibodies, resulting in their remission13. Biotech companies such as Cabaletta Bio or Kyverna Therapeutics as well as pharma giant, Novartis are now exploring the potential of CAR-T technology in other immune-mediated inflammatory disorders such as mucosal pemphigus vulgaris (mPV), myositis or scleroderma. In addition, while traditional CAR-T cell therapies are based on effector T cells, Sangamo Therapeutics is currently evaluating the efficacy of regulatory T cells, or Tregs, in preventing graft rejection in patients who have undergone kidney transplantation. Positive results from this trial could open new avenues of treatment for other autoimmune diseases, such as multiple sclerosis, inflammatory bowel disease, rheumatoid arthritis, or type 1 diabetes.
Despite recent progress in the development of adoptive cell therapies, the initial advances in the field came through using undifferentiated or in vitro differentiated stem cells for the treatment of degenerative and systemic conditions. In addition to neurological and neuromuscular diseases such as Alzheimer’s, Parkinson’s, and Muscular Dystrophy, the use of various multi- or pluripotent stem cells has also been shown experimentally to aid in combatting loss of function from physical injuries or heritable autoimmune diseases. Current approved stem-cell therapies include the use of mesenchymal stem cells (MSCs), hematopoietic stem cells (HSCs), or induced pluripotent stem cells (iPSCs).
MSCs are multipotent cells derived from sources such as umbilical cord blood, amniotic fluid, or bone marrow. MSC therapies have been applied as a treatment for a wide variety of diseases, including cardiovascular diseases, degenerative disorders, and inflammatory bowel diseases. MSCs from cord blood could be used to treat Type 2 diabetes and slow the loss of pancreatic insulin production14, or to selectively migrate to areas of brain injury and repair damaged brain tissue, as observed in cerebral palsy.15 Cord blood stem cells are strong candidates for such regenerative therapies due to their autologous nature, which reduces the risk of exhibiting graft versus host disease.
HSCs are multipotent stem cells able to self-renew and differentiate into mature blood cells that form the myeloid and lymphoid cell lineages. They are generally bone marrow-derived, but many successful products have also utilized cord blood sources due to their tolerance for HLA-type mismatches between donors and patients and the avoidance of graft versus host disease. HSC products have already been approved for the treatment of blood disorders through transplantation to reconstitute the hematopoietic and immunologic systems.16 This year, a new allogeneic product derived from cord blood hematopoietic progenitor cell cells, Omidubicel from Gamida Cell, was approved for the treatment of hematological malignancies and another one, a HPC cord blood from StemCyte anticipates licensure later in 2023.4
iPSCs are adult cells derived from skin or blood tissue that have been reprogrammed back into an embryonic-like pluripotent state. This, in turn, generates a self-renewing cell source that can be directed in vitro into any type of human cell required for therapeutic purposes. For example, to treat hematopoietic disorders, musculoskeletal injuries, spinal cord injuries, and liver damage by generating specific cells.17 Together, stem cells provide immense opportunities for cell therapies to be applied in previously untreatable conditions.
Prospects, Promises, and Risks of Cell Therapy
The application and development of cell therapy technologies carry immense potential, but also a lot of risk. These risks lie not only in patient safety but also for pharmaceutical companies, and in return on investment following a very nuanced, complex, and costly development process that can vary widely from one company to another. Mitigating risks and maximizing efficacy are critical to the success of any new therapeutic product, however, cell and gene therapies carry with them the added cost of personalized development. This added product development cost, especially that of autologous cell therapies, demands a continuous examination of processes to maximize efficiency. Given the staggering speed at which these technologies change, it is evident that companies operating in this field require flexible partnerships with collaborators, CROs, and vendors. Valuable qualities of these organizations include a comprehensive knowledge base and the agility to evolve in concert with the changing needs of partner companies. Around 3000 clinical trials are currently underway for cell and gene therapies1 and adapting organizational infrastructure to monitor the tremendous amounts of scientific and clinical information and enable real-time decision-making is essential.
Cell therapies could provide effective treatment solutions for several diseases, but it is only through gaining a clear understanding of the science behind each new methodology that we can fully determine the breadth of what each therapy can offer a patient and how to ensure the best healthcare outcome possible. This understanding is also required to continue making technological advances in cell therapy that will enable its application in the treatment of a wider variety of diseases beyond cancer. When developing these therapies, it is important to understand the background and characteristics that differ from patient to patient (and donor to donor) to match the right therapy to the right patient in a precise manner.
Maximum precision can only be achieved from these therapies using a technological solution that assesses outcomes by correlating product efficacy, patients stratified by suitability for a product, and markers of treatment success. Genedata Profiler® is an innovative software solution that allows users to overcome the complex challenges faced during the development and administration of cell therapy products. It facilitates the efficient integration of voluminous real-time data to track efficacy, maintenance of desired cellular phenotypes, and population expansion. The capability to harmonize layered multi-modal data while enabling patient stratification is crucial in the development of cell therapies. It allows the acceleration of key decisions in the development of new methods and products, whilst increasing manufacturing process efficiency, reducing costs, and treatment delivery time. Genedata is committed to supporting companies framing the future of cell and gene therapy by improving the efficiency of processes and thereby ensuring the best care for patients before, during, and following treatment.
Learn More About Genedata Profiler
Authors:
Nicholas S. Geraci, Ph.D., Scientific Consultant, Genedata Profiler
Justyna Lisowska, Ph.D., Scientific Marketing Manager, Genedata Profiler
References:
- American Society of Cell and Gene Therapy (ASGCT). Gene, Cell & RNA Therapy Landscape Q1 2023 Quarterly Data Report. 2023
- Guedan S, et al. Emerging cellular therapies for cancer. Annual Review of Immunology. 2019.
- Stevanović S, et al. The landscape of immunogenic tumor antigens in successful immunotherapy of virally induced epithelial cancer. Science. 2017.
- Johnson V. and Bigica A. 10 Cell and Gene Therapies to Watch in 2023. CGT live. 2023
- Rosenber SA, et al. Adoptive cell transfer: a clinical path to effective cancer immunotherapy. Nature Reviews Cancer. 2008.
- Gettinger S, et al. Impaired HLA class I antigen processing and presentation as a mechanism of acquired resistance to immune checkpoint inhibitors in lung cancer. Cancer Discovery. 2017.
- June CH, et al. Chimeric Antigen Receptor Therapy. The New England Journal of Medicine. 2018.
- Ahmed N, et al. Human epidermal growth factor receptor 2 (HER2)–specific chimeric antigen receptor-modified T cells for the immunotherapy of HER2-positive sarcoma. Journal of Clinical Oncology. 2015.
- Brown C. E, et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. New England Journal of Medicine. 2016.
- Liu S, et al. NK cell-based cancer immunotherapy: from basic biology to clinical development. Journal of Hematology & Oncology. 2021.
- Ginefra P, et al. Innate Immune Cells and Their Contribution to T-Cell-Based Immunotherapy. Int J Mol Sci. 2020
- Kather JN, et al. Harnessing the innate immune system and local immunological microenvironment to treat colorectal cancer. Britsh Journal of Cancer. 2019.
- Gomes A, et al. Human umbilical cord mesenchymal stem cells in type 2 diabetes mellitus: the emerging therapeutic approach. Cell and Tissue Research. 2021.
- Dong H, et al. Umbilical cord mesenchymal stem cell (UC-MSC) transplantations for cerebral palsy. American Journal of Translational Research. 2018.
- Wang LL, et al. Cell therapies in the clinic. Bioengineering and Translational Medicine. 2021.
- Singh VK, et al. Induced pluripotent stem cells: applications in regenerative medicine, disease modeling, and drug discovery. Frontiers in Cell and Developmental Biology. 2015.