Accelerating Gene Therapy R&D
The leading workflow platform for increasing the efficiency of gene therapy R&D, Genedata Biologics® supports diverse viral delivery systems including adeno-associated virus (AAV), adenovirus, and retrovirus vectors.

AAVs & Gene Therapies
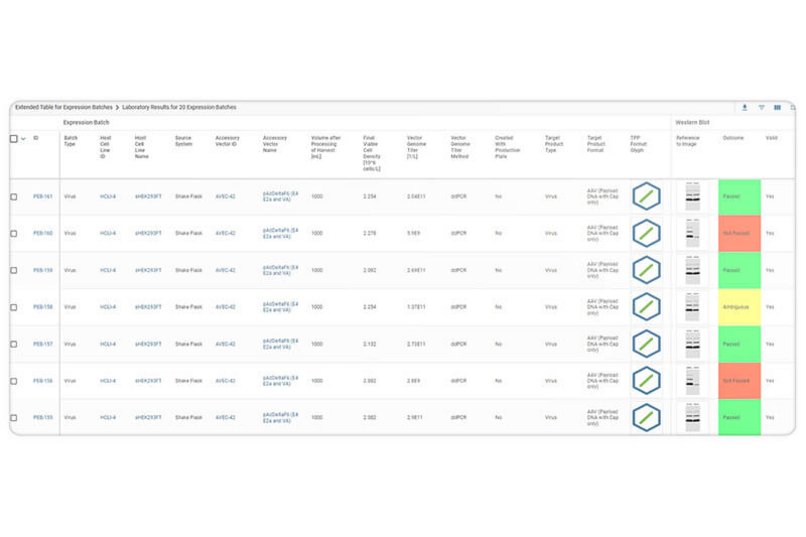
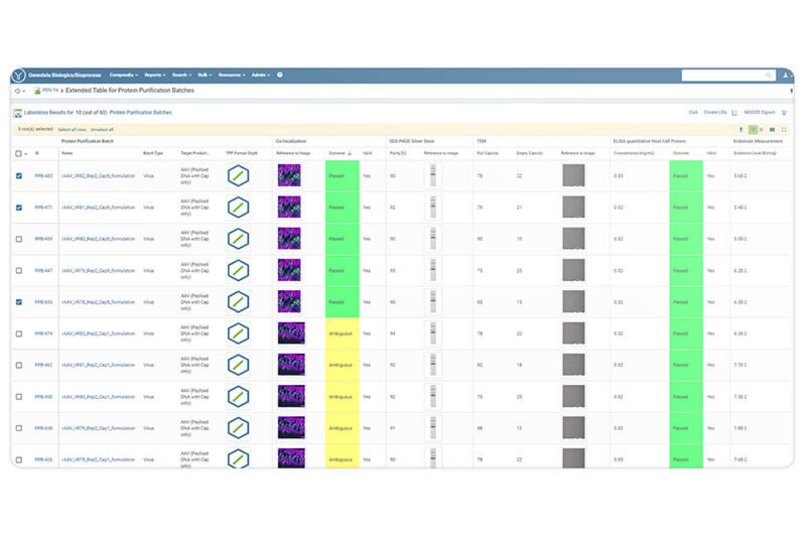
Genedata Biologics covers the entire gene therapy workflow, from serotype optimization and viral vector design to virus packaging, purification, and testing. It tracks all samples and associated information including customized vectors and packaging plasmids, cell lysates, purified virus batches and their sequences, titers, and assay results such as empty-to-full capsid ratios. Genedata Biologics provides valuable, real-time insights leading to improved quality control, assay standardization, and more accurate viral potency assessments, putting all critical decision factors in one place to facilitate scale-up and reproducibility during vector manufacturing.
Customer Stories

Accelerating AAV-Based Gene Therapy Development
“Leveraging Genedata’s capabilities will enable us to continue scaling Encoded’s process development capabilities to drive growth and value in gene therapy."