Bioprocess Development. Next-Generation.
Genedata Bioprocess® designs next-generation bio-manufacturing processes across CLD, USP and DSP, formulation, and analytics development. It enforces data integrity and compliance, streamlining development of originator drugs and biosimilars.
Purpose-Built. Digitalizing
Next-Generation Process Design.
Achieve operational excellence with the only platform uniquely designed from the start for the development of biotherapeutic drugs. Acting as an integrated data backbone, Genedata Bioprocess ensures maximal efficiency along the whole end-to-end workflow and supports cutting-edge technologies, such as highly parallel scale-down bioreactor panels, as well as complex DSP unit operations and sophisticated analytical techniques.
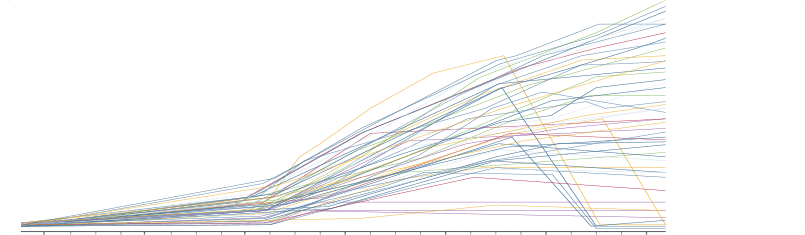
End-to-End Platform.
Highest Data Volumes.
Pharmaceutical giants, as well as renowned CMOs around the world, use Genedata Bioprocess to increase the efficiency of their development processes. Scaling with highest data volumes, the platform integrates and harmonizes overall bioprocess development and CMC workflows, streamlines communication and handovers, and provides central and transparent real-time access to all process information.
Automate & Industrialize.
Improve Productivity.
By directly integrating with all laboratory instruments, such as bioreactors, downstream processing equipment, and analytics devices, Genedata Bioprocess automates data capture, which dramatically improves overall productivity and reduces manual errors. The platform can also be integrated with robotics equipment, enabling full automation and high-throughput processes, such as HT formulation screens.
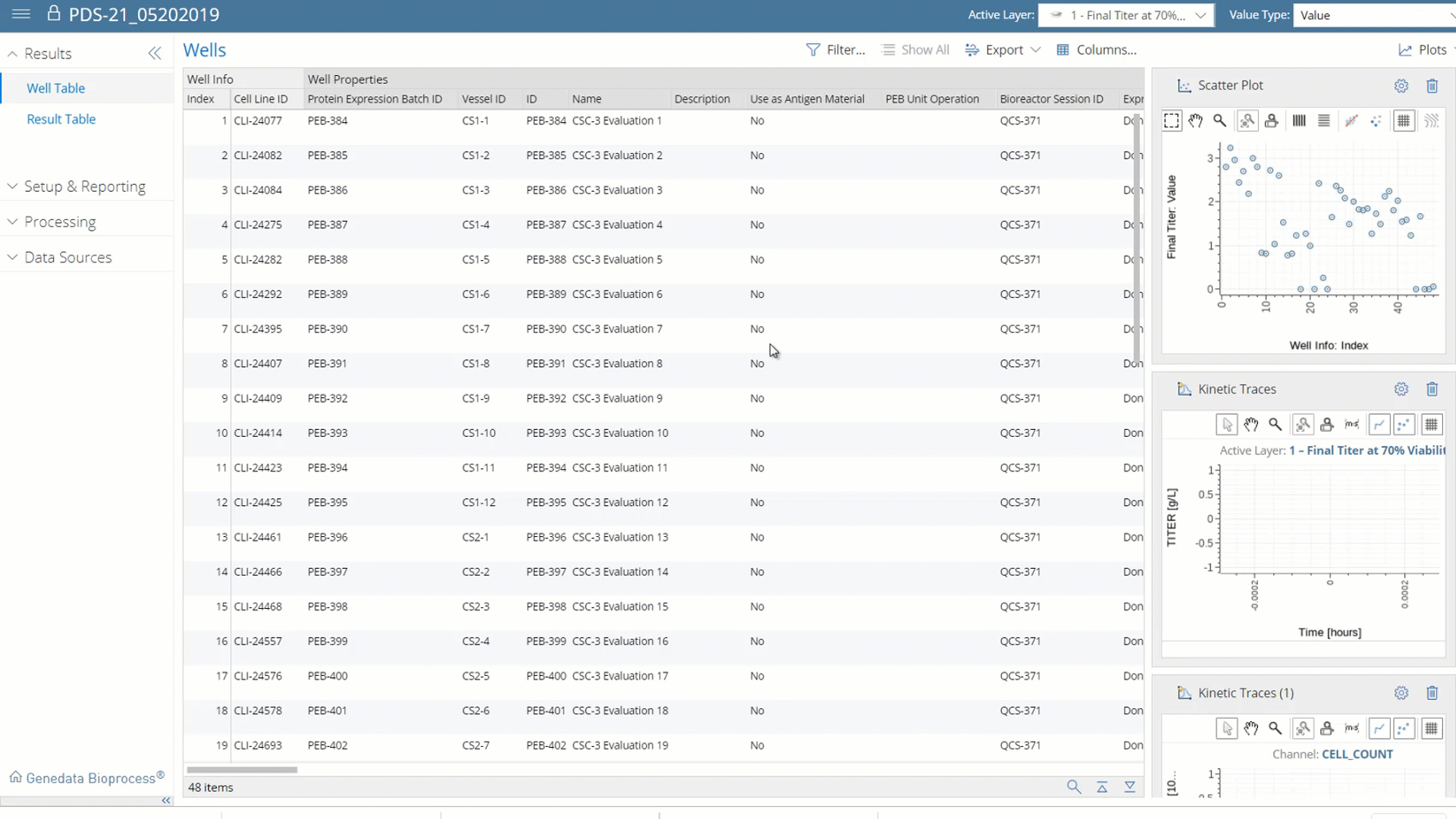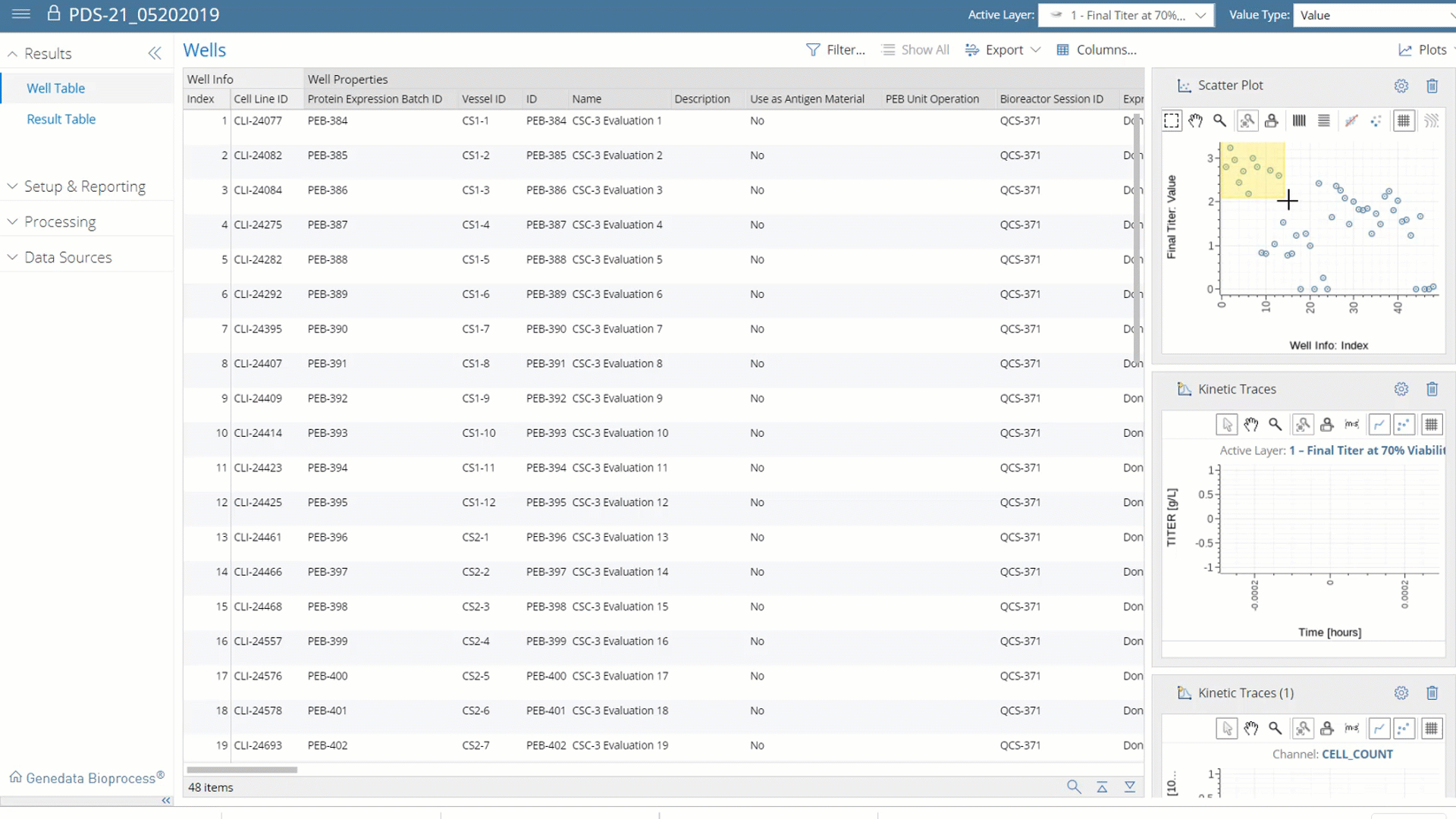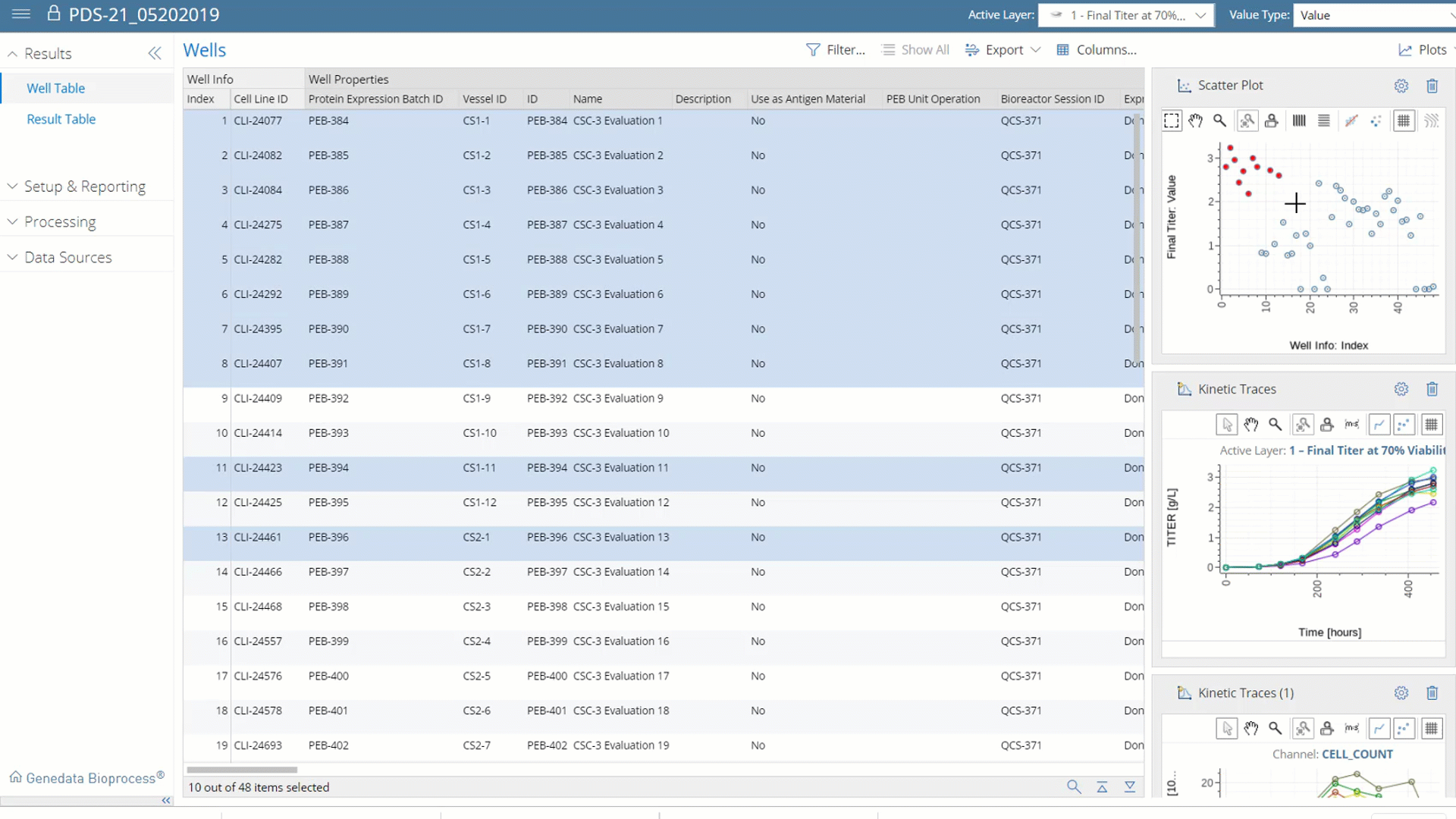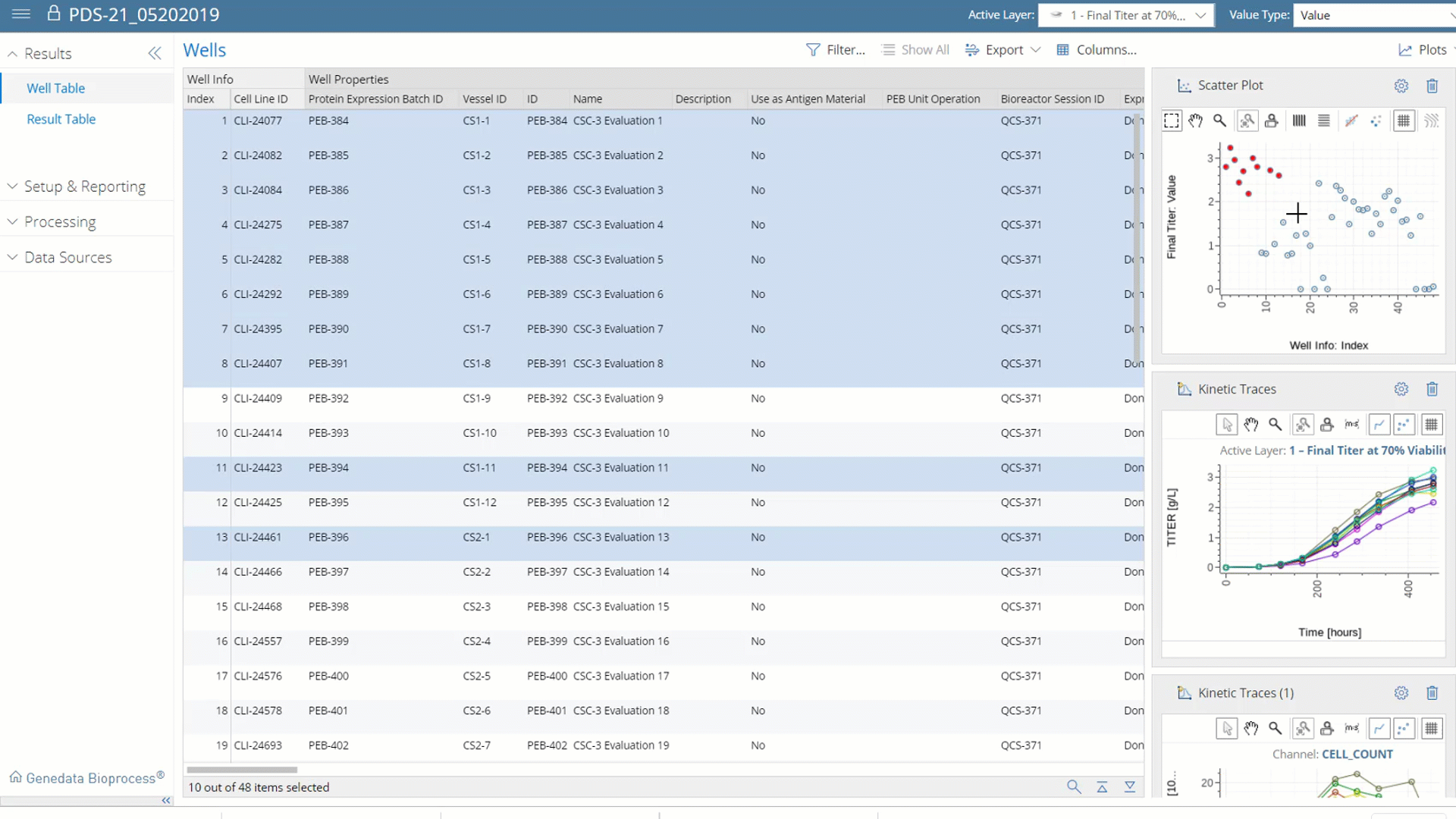
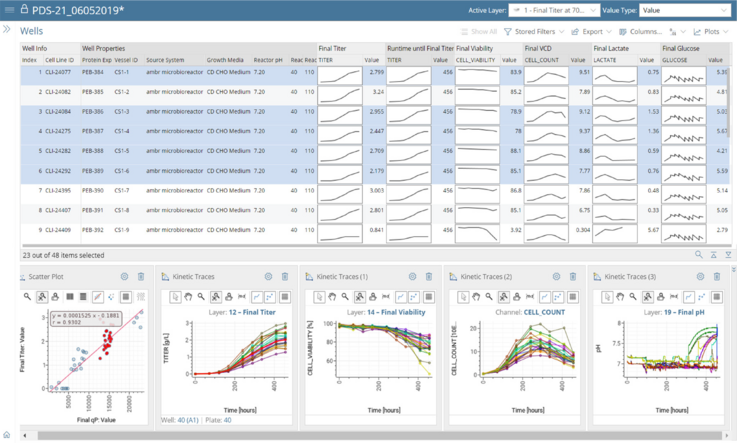
More than Data Capture.
Data-Driven Decision Making.
With Genedata Bioprocess, data is centrally captured, structured, and stored, which makes it possible, for the first time, to fully exploit its value by analyzing all data both within and across projects. This enables a systematic Quality-by-Design (QbD) approach that identifies and monitors product critical quality attributes (CQAs) and correlates these with critical process parameters (CPP). Cross-project data investigations guide process and technology enhancements to eliminate workflow bottlenecks.

Ensure Data Integrity.
Eliminate Compliance Risks.
Acting as a central data backbone, Genedata Bioprocess automatically documents the full workflow. The platform’s audit trail functionalities allow you to easily trace back all data along the full process, which ensures data integrity and regulatory compliance for every step along the workflow. Genedata Bioprocess can be used either in a fully compliant or non-compliant mode.