Identifying the Root Cause of An Unusual Biological Product Characteristic using Next-Generation Sequencing
March 10, 2022
Marie-Ange Kouassi
Biological molecules provide promising treatment options allowing to alter disease mechanisms with precision. Yet, the development of these molecules can be challenging. During the development process, it is critical to check for changes that may occur and affect the functionality, safety, and efficacy of these therapeutic products.
Successful cell line development requires routine screening to ensure continuous generation of the desired product. As these biological molecules are purposely intended as treatment, it is critical to ensure they do not change in a way that makes them ineffective in targeting disease, or detrimental to patient health. By screening production cell lines at Boehringer Ingelheim, Fischer et al. identified an unusual characteristic of monoclonal antibodies produced by their proprietary Chinese hamster ovary (CHO) cells. The antibody generated in one cell line exhibited a high level of sialylation with NGNA (N-glycolylneuraminic acid) instead of the typical NANA (N-acetylneuraminic acid). Knowing this could have adverse effects in patients by inducing an elevated immunogenic response, the team decided to investigate the reason for this unusual phenotype. They used Next-Generation Sequencing (NGS) applications to delve deep into the root cause and identified a new microRNA (miR-111). They also identified a single point mutation that altered a binding site allowing the binding of a different transcription factor and causing the downregulation of miR-111. The lack of miR-111 allowed an unusual expression of cytidine monophosphate-N‐acetylneuraminic acid hydroxylase (CMAH), leading to increased NGNA antibody sialylation.
To investigate the phenotypic differences between the two clones (one with normal NANA sialylation and another with high NGNA sialylation), Fischer et al. generated a cell-specific reference genome knowing its importance in serving as an accurate reference for all sequencing data analysis performed. Genedata Selector® was used to assemble and annotate the CHO cell genome which was enhanced with genomic DNA and RNA data specific to Boehringer Ingelheim’s cell line. Following NGS, mutation profiles were generated, and gene expression patterns were analyzed. Advanced statistical analyses (e.g., principal component and correlation analyses) were performed using Genedata Selector to obtain a deeper biological understanding of the underlying data.
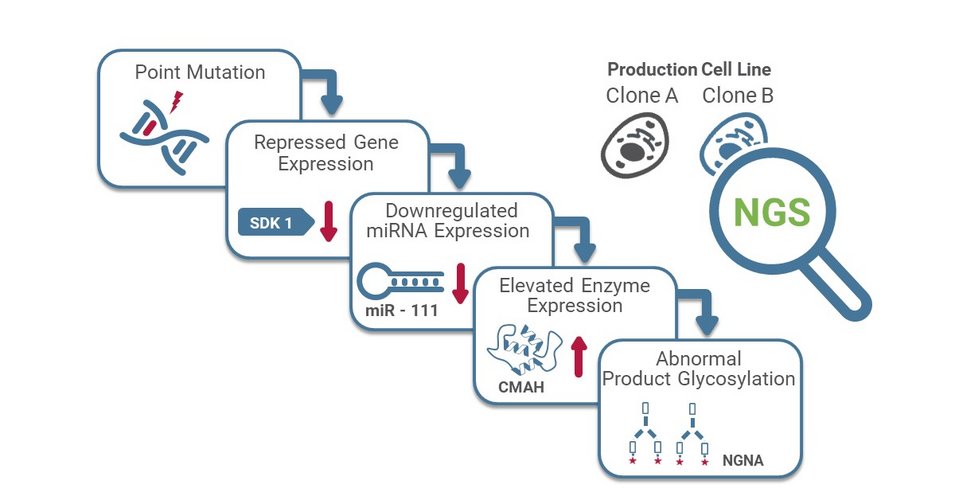
From the results generated, Fisher et al. identified approximately 1990 differentially expressed genes between the two clones. One of them was for the enzyme cytidine monophosphate-N-acetylneuraminic acid hydroxylase (CMAH) which converts CMP-NANA to CMP-NGNA, the precursor of NGNA. The team then used CRISPR Cas9 technology to knock out the catalytic activity of the CMAH gene in a derived cell line leveraging Genedata Selector for designing single-guide RNAs. This interruption resulted in a lost sialylation of the secreted antibodies confirming that the activity of CMAH was indeed causing this abnormal phenotype. One potential hypothesis was that CMAH is normally repressed by a regulatory microRNA and due to the loss of its expression, CMAH expression and activity were upregulated. After performing a differential gene expression analysis specific for small non-coding RNAs, one of the downregulated candidates identified was the microRNA, miR-111. An anticorrelated expression observed between CMAH and this newly identified microRNA named miR-111 led the team to look further into the loss of miR-111 expression.
By predicting the transcriptional binding sites using algorithms implemented in Genedata Selector, it was demonstrated that a binding site for miR-111 was present on CMAH. This was functionally validated in experiments where miR-111 function was restored in the clone producing antibodies with high NGNA sialylation. Following transfection, CMAH expression was downregulated in the clones, leading to a reduced NGNA sialylation proving that miR-111 regulates CMAH and NGNA sialylation. Fisher et al. then explored the genome sequencing data to understand how miR-111 expression was lost. It was identified that miR-111 was located in an intron of a gene named SDK1. Driven by the same promoter, both miR-111 and SDK1 expression was lost in the clone with high NGNA sialylation.
The team identified a single point mutation in the promoter region of SDK1 which made the binding site complementary to HINF-P, a highly expressed repressor in CHO cells. This repression of SDK1 also repressed miR-111 causing upregulated CMAH expression and increased NGNA sialylation. This study proved that changes in the expression of a newly discovered microRNA, miR-111, altered the catalytic activity of a key enzyme, CMAH which led to the abnormal attachment of a sialic acid residue, NGNA, on monoclonal antibodies produced by CHO cells. Through this omics-assisted cell line characterization study, the authors demonstrated that NGS is a powerful method for troubleshooting during cell line development for recombinant protein production.
To find out more, read the publication, or watch the talk delivered by the first author of this paper, Simon Fischer (Ph.D.). In the webinar he explains in detail how his team at Boehringer Ingelheim interrogated the differences between the two clones by leveraging NGS for troubleshooting. Simon is a Senior Principal Scientist and Head of Cell Biology at Boehringer Ingelheim and presented this talk at our last biosafety Open Forum.